How Does The Cell Know To Make Pfk And Hexokinase
5.i: Glycolysis
- Folio ID
- 16114
Nearly all metabolic reactions are catalyzed by enzymes in club to go on upwards with the energy and cloth demands of the cell. In fact, the discussion of some of the metabolic processes in this chapter will well-nigh seem to be laundry lists of enzymes. We volition brainstorm with one such list in describing the catabolism of the simple sugar, glucose, through the process of glycolysis.
Glycolysis
Whether the cell is prokaryotic or eukaryotic, one of its bones methods for generating usable energy is glycolysis. This process uses glucose, which is the most common free energy source for most cells. Even so, glucose cannot be directly cleaved down to provide free energy for the cell: glycolysis is a process that breaks it down in a series of reactions to create adenosine triphosphate (ATP), which is the most common energy "currency" of the cell. That is, ATP can release usable free energy in a single reaction.
Glucose, being a 6-carbon sugar, has a large amount of potential energy stored in its bonds. However, since it is thermodynamically stable, it would have the investment of a lot of external energy to release the energy of glucose in one step (e.chiliad. lighting it on fire to break it down into CO2 and HiiO), and non only is information technology impossible for cells to generate that kind of energy at one time, the cell has no mechanism to use all the energy released at one instant in time. Nearly of it would be wasted as excess heat. Instead, the cell uses enzymes to destabilize and break down the sugar through a series of conversions into intermediate compounds. The bones procedure and enzymes involved are as follows.
i. Glucose is phosphorylated past hexokinase to make Glucose-6-Phosphate. The enzyme is so named considering information technology is a kinase (puts a phosphate group on) that acts on a hexose (six-carbon sugar). In this case, it places the phosphate on the 6-carbon of glucose. Nevertheless, hexokinase can too phosphorylate other hexoses such as fructose and mannose (all in the D-conformation). There are ii major reasons this is good for the prison cell. Since glucose concentration is higher inside the prison cell than outside, there is pressure level for information technology to movement dorsum out of the cell. By converting it to G6P, it is no longer role of the glucose concentration gradient, and it has a charged phosphate group, making information technology well-nigh impossible to leak out of the membrane. The improver of the phosphate also increases the energy in the molecule, making information technology less thermodynamically stable, so that it can exist broken downwards. This reaction requires the use of ATP as a phosphate donor and the energy needed to adhere it. That is, free energy is used in this footstep, non produced. Consider it an investment of energy though, since by the end of glycolysis, more ATP is produced than used.
Hexokinase requires ATP in the form of a complex (to the twond and 3rd phosphate groups) with a divalent cation, typically Mg2+ in vivo. ATP alone is actually a competitive inhibitor of hexokinase. The product, G6P, as well functions as an inhibitor, thus providing some measure of feedback regulation. In fact, musculus cells using glycogen stores catechumen the glycogen directly to G6P, so hexokinase activity is very low in those cells.
2. Glucose-6-Phosphate is converted to Fructose-6-Phosphate by phosphoglucose isomerase. As the proper noun implies, the isomerase merely rearranges the existing atoms within the G6P to brand the F6P without removal or addition of whatever atoms.
3. Fructose-6-Phosphate is phosphorylated by phosphofructokinase (PFK) to Fructose- 1,6-bisphosphate. There is again an investment of an ATP to provide the phosphate group and the energy to attach information technology.
PFK is an important regulator of glycolysis. It is a tetrameric protein, and each subunit has two bounden sites for ATP: one is the normal substrate site, the other is an inhibitory site such that binding of ATP lowers the enzyme's analogousness for F6P. ATP is not the only regulator of PFK activity: AMP is also a positive regulator of PFK, and can increment information technology up to v-fold.
4. The Fructose-1,half-dozen-bisphosphate is cut in half by aldolase, yielding a molecule of dihy- droxyacetone phosphate and a molecule of glyceraldehyde-3-phosphate.
There are two classes of aldolases: class I are found in animals and plants, while class II are plant in fungi and leaner. Class I require no cofactors, but course II require a divalent cation (physiologically usually Fe2+ or Zn2+).
5. The G3P can participate in the next reaction, but the dihydroxyacetone phosphate, despite its similarity, cannot. And then, it needs to be rearranged by triose phosphate isomerase, which converts it to another molecule of glyceraldehyde-3-phosphate.
Triose phosphate isomerase is a "perfect enzyme" that catalyzes the formation of product equally fast as the enzyme and substrate tin can brand contact in solution (i.e. rate is purely improvidence-limited).
6. Each of the two molecules of G3P generated from the glucose molecule now undergo oxidation catalyzed by glyceraldehyde-3-phosphate dehydrogenase (GAPDH) in the presence of NAD+ and inorganic phosphate (Pi). Each of these reactions produces 1,three-bisphosphoglycerate, which has a high-energy phosphate group, and NADH. NADH is a loftier free energy electron carrier (electron comes from G3P). In eukaryotes with an
aerobic environs, this NADH will probable be used to aid generate ATP through the tricarboxylic acrid bicycle (aka Krebs cycle or citric acid cycle). In anaerobic situations, the NADH will participate in fermentation for reasons discussed in the side by side section.
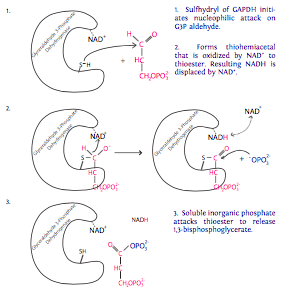
7. The phosphate grouping on the 1-carbon of 1,3-bisphosphoglycerate is transferred to ADP past phosphoglycerate kinase to make 3-phosphoglycerate and ATP (finally!). From the two molecules of G3P entering pace 6, we become two molecules of ATP to provide energy for the cell in this pace. Recalling the earlier investment of ATP (in steps i and 3), the reaction has merely "broken fifty-fifty" at this point. two in, 2 out.
The name of the enzyme suggests that a phosphate is added to phosphoglycerate. This is not a error: recall that enzymes can catalyze reactions in either direction, depending on reaction weather condition. Nether weather condition of loftier phosphoglycerate and ATP, phosphorylation of phosphoglycerate would occur. However, the physiological conditions are a relatively loftier concentration of the 1,iii-bisphosphoglycerate in comparison to relatively depression levels of phosphoglycerate thus driving the reaction "backwards" with respect to the naming of the enzyme.
8. The 3-phosphoglycerate is then rearranged by phosphoglycerate mutase to make 2-phosphoglycerate. This molecule has a higher complimentary energy of hydrolysis than when the phosphate grouping is on the 3-carbon.
The action of phosphoglycerate mutase is non just the intramolecular phosphate group transfer that information technology seems to exist at first glance. The enzyme must first be activated by phosphorylation, and it is the enzyme's phosphate that is added to the 2-carbon of 3PG. The doubly-phosphorylated intermediate so transfers its 3-phosphate to the enzyme, and 2PG is released.
nine. That energy is used to create ATP, as the two-phosphoglycerate undergoes dehydration by enolase to brand phosphoenolpyruvate (PEP).
PEP is made because hydrolysis of the phosphate from 2PG does not release enough energy to drive phosphorylation of ADP to ATP. PEP hydrolysis, on the other mitt, releases significantly more than needed.
10. Pyruvate kinase and so transfers a loftier energy phosphate grouping from PEP to ADP, producing an ATP for utilise by the cell, and pyruvate.
Pyruvate kinase requires not only divalent Mgii+ as with most other kinases, but besides K+. The enzyme works in ii steps: the ADP attacks the PEP phosphorus to make ATP and enolpyruvate. Enolpyruvate is then converted to its keto tautomer.
Keeping in heed the doubling of reactions from steps 6-10 (splitting of fructose-1,6- bisphosphate generates 2 G3P), the total usable energy production from glycolysis of a single molecule of glucose is 4 ATP and 2 NADH. However, the net ATP production is but 2 ATP if we remember the initial investment of two ATP in the early steps. Not really anything to write home about. Furthermore, although the NADH and pyruvate can participate in the tricarboxylic acid wheel in aerobic eukaryotic situations to generate a significant amount of ATP, in anaerobic situations, they practice not produce usable energy.

Bidirectional arrows indicate enzymes used for both glycolysis and gluconeogenesis. Unidirectional arrows indicate enzymes that only function in glycolysis. *Note that reactions 6-x are occurring in indistinguishable (two G3P from one glucose).
Thus anaerobic ATP production, i.due east. glycolysis, is far less efficient at extracting free energy from a glucose molecule than aerobic ATP production, which can generate approximately 38 ATP per glucose. On the other hand, when a lot of ATP must be generated rapidly, glycolysis is the mechanism of pick, in cells such every bit the fast-twitch fibers of skeletal muscle. These cells really accept very few mitochondria considering glycolysis can produce ATP at a much college (upward to 100 times) rate than oxidative phosphorylation. What happens to the pyruvate and NADH? In aerobically metabolizing cells, they get to the mitochondria for the TCA cycle and oxidative phosphorylation. In anaerobes, they undergo fermentation.
Note that the NADH produced by glycolysis in the cytoplasm does not directly participate in oxidative phosphorylation in the mitochondria since the inner mitochondrial membrane is impermeable to it, but it sends a "virtual equivalent" into the mitochondria via ane of ii pathways: the aspartate-malate shuttle combines malate-α-ketoglutarate antiports, aspartate-glutamate antiports, and metabolite interconversion by transaminase with malate dehydrogenase to oxidize NADH cytoplasmically and use the energy generated to reduce NAD+ in the mitochondrial matrix; the other pathway is a DHAP shuttle system, in which NADH is used to reduce dihydroxyacetone phosphate to glycerol-iii-P using a cytoplasmic glycerol-3-phosphate dehydrogenase, and the cycling the DHAP to glycerol-iii-P via a flavoprotein dehydrogenase embedded in the inner mitochondrial membrane. This flavoprotein dehydrogenase takes the electrons from glycerol-three-P to brand FADHtwo, which can participate in the electron send concatenation.
The DHAP or glycerophosphate shuttle is less efficient than the malate-aspartate shuttle, generating approximately two ATP vs 2.7 ATP per NADH. All the same, information technology tin can operate even when the concentration of cytoplasmic NADH is low, as happens in tissues/cells with a very loftier metabolic rate (including skeletal muscle and brain), while the malate-aspartate shuttle (prevalent in liver and center) is sensitive to the relative concentration of NADH and NAD+.
Source: https://bio.libretexts.org/Bookshelves/Cell_and_Molecular_Biology/Book:_Cells_-_Molecules_and_Mechanisms_%28Wong%29/05:_Metabolism_I__Catabolic_Reactions/5.01:_Glycolysis

0 Response to "How Does The Cell Know To Make Pfk And Hexokinase"
Post a Comment